The College adopted the Model Standards for Pharmacy Compounding of Non-Sterile Preparations and the accompanying Guidance Document for Pharmacy Compounding of Non-Sterile Preparations, following approval at the December 2017 Board meeting, with full implementation by January 1, 2022.
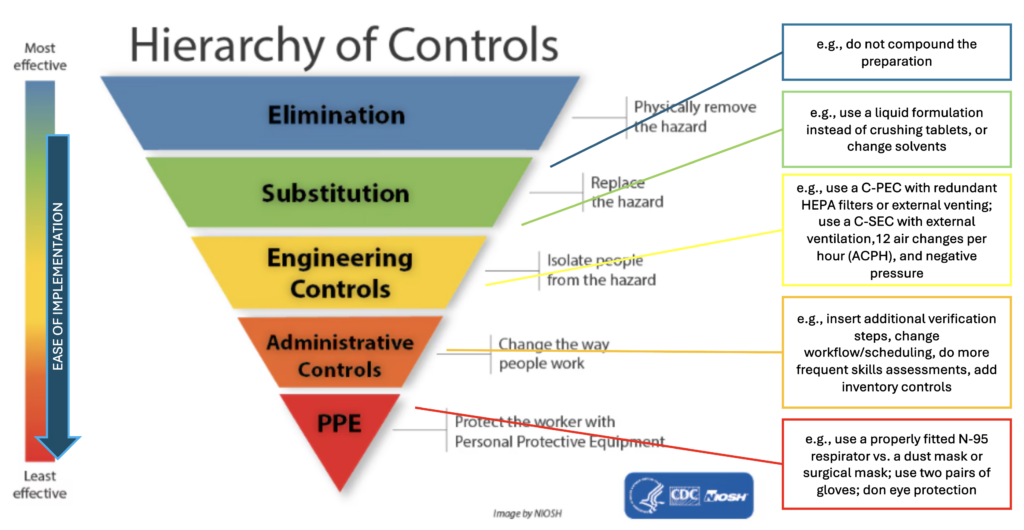
The standards apply to all pharmacy professionals in all practice settings that engage in non-sterile compounding in any capacity and are an important way to ensure patient safety and protect pharmacy personnel.
The Designated Manager (in a community pharmacy) or pharmacy department head (hospital pharmacy) must ensure, with the best interest of the patient in mind, that the pharmacy has the resources necessary to compound a preparation of good quality in a safe and appropriate manner. When this is not possible, they have an ethical obligation to make reasonable efforts to ensure continuity of patient care.
UPDATE: Following the principles of right-touch regulation and approval from the Board in March 2024, the College has developed a mandatory training course for new compounding supervisors in all pharmacies and current compounding supervisors of pharmacies where standards are not being met.
Access all Pharmacy Connection non-sterile compounding articles.